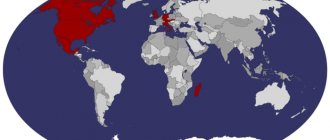
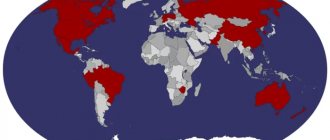
Probably the first association with the word “platinum” will be something like a platinum wedding ring, or any expensive jewelry with a diamond. As a last resort, a platinum commemorative coin, or an investment deposit. However, in real life the situation is different. Thus, platinum is one of those precious metals whose industrial use is more common than its use for jewelry.
According to USGS data for 2012
Story
The inhabitants of Ancient Egypt were the first to become acquainted with metal: archaeologists unearthed jewelry that was nearly three thousand years old. A little later, the Indians of South America began to make platinum artifacts.
Platinum nugget, Conder deposit
Europe
The Spanish conquistadors who conquered America in the 16th century also found platinum veins. But they mistook the metal for silver, and even “defective”: it did not want to melt. The new metal was sold at half the price of silver bars.
And they came up with the appropriate name - platina in Spanish means “small silver, little silver” (from plata - silver).
Everything changed when Spanish jewelers managed to fuse “silver” with gold:
- The first counterfeits of gold appeared. It was not possible to discover this for a long time due to the density of platinum.
- When the deception was discovered, the import of platinum was banned by decree of the king, and all the “cash” was drowned in the sea.
- In 1751, the Swedish scientist Theophile Schaeffer, who examined the metal, discovered its preciousness.
- Since 1778, the new metal began to be used as a self-sufficient jewelry material.
- In 1870, the ruler of France, Louis XVI, considered platinum the only royal metal.
The Second World War “closed” platinum for civilian consumption: the military took everything.
Russia
In Russia, the history of platinum began in 1819, when fragments of the “new Siberian metal” were discovered in alluvial gold in the Urals.
In the 19th century, Russia provided 95% of the world's platinum production.
The local population used silver-steel grains instead of shot for hunting. The first domestically produced platinum products were a ring and a teaspoon. Their craftsmen presented them to Emperor Alexander I.
Coin 3 rubles, 1834
They didn’t know what to do with platinum. They minted a coin (3, 6, 12 rubles), but its cost exceeded the face value many times over. As a result, money was withdrawn from circulation, and the British bought coins as raw materials.
Today, almost everything that is mined in Russia remains in the country.
Jewelry world
The global platinum rush began at the end of the second millennium:
- 1930s – fashion for metal was introduced by style icon Marlene Dietrich. Soon the epidemic spread to America.
- Edward VII and Wallis Simpson became engaged with platinum rings from Cartier, then Elvis Presley and Priscilla Ann.
- It was finally elevated by the first-tier jewelry brand Tiffani.
It has been Japan's favorite metal since the 1960s - its white color symbolizing the core virtues of this community. Two-thirds of the world's jewelry is purchased by China.
Interesting Facts
- The largest platinum nugget currently existing is the “Ural Giant” weighing 7,860.5. Kept in the Diamond Fund of the Moscow Kremlin.
- In South America in the 17th century, platinum was considered “fake silver” and one day its reserves were drowned in the ocean to prevent counterfeiting.
- The world's first platinum coins were issued in Russia (see Platinum coins).
- In Isaac Asimov's short story series "I, Robot" and his other works, the positronic brains of robots are made of spongy platinum (more precisely, an alloy of platinum and iridium).
Physico-chemical characteristics
It is the heaviest, most durable of the precious metals. Does not oxidize, does not darken, does not rust. Interacts poorly with other substances, even aggressive ones. Refractory, but plastic, viscous.
Platinum crystals
Only “royal vodka” can dissolve it, and not immediately.
| Properties of the atom | |
| Name, symbol, number | Platinum (Pt), 78 |
| Atomic mass (molar mass) | 195.084 a. e.m. (g/mol) |
| Electronic configuration | [Xe] 4f14 5d9 6s1 |
| Atomic radius | 139 pm |
| Chemical properties | |
| Covalent radius | 130 pm |
| Ion radius | (+4e) 65 (+2e) 80 pm |
| Electronegativity | 2.28 (Pauling scale) |
| Electrode potential | Pt←Pt2+ 1.20 V |
| Oxidation states | 4, 2, 0 |
| Ionization energy (first electron) | 868.1 (9.00) kJ/mol (eV) |
| Thermodynamic properties of a simple substance | |
| Density (at normal conditions) | 21.09-21.45 g/cm³ |
| Melting temperature | 2041.4 K (1768.3 °C, 3214.9 °F) |
| Boiling temperature | 4098 K (3825 °C, 6917 °F) |
| Ud. heat of fusion | 21.76 kJ/mol |
| Ud. heat of vaporization | ~470 kJ/mol |
| Molar heat capacity | 25.8 J/(K mol) |
| Molar volume | 9.10 cm³/mol |
| Crystal lattice of a simple substance | |
| Lattice structure | cubic face-centered |
| Lattice parameters | 3.920 Å |
| Debye temperature | 230.00 K |
| Other characteristics | |
| Thermal conductivity | (300 K) 71.6 W/(mK) |
| Thermal expansion | (25 °C) 8.8 |
| Young's modulus | 168 GPa |
| Shear modulus | 61 GPa |
| Volume control module | 230 GPa |
| Poisson's ratio | 0,38 |
| Mohs hardness | 3,5 |
| Vickers hardness | 549 MPa |
| Brinell hardness | 392 MPa |
| CAS number | 7440-06-4 |
Iridium, palladium, rhodium, ruthenium, and osmium have similar physical and chemical properties and a cubic structure. These metals are grouped into the platinum group.
Being in nature
Isotopes
Natural platinum occurs as a mixture of six isotopes: 190Pt (0.014%), 192Pt (0.782%), 194Pt (32.967%), 195Pt (33.832%), 196Pt (25.242%), 198Pt (7.163%). One of them is weakly radioactive (190Pt, alpha decay in 186Os, half-life 6.5⋅1011 years). The existence of very weak radioactivity of two more natural isotopes of platinum is predicted: the alpha decay 192Pt→188Os and the double beta decay 198Pt→198Hg, however, these decays have not yet been experimentally recorded; it has only been established that the half-lives exceed 4.7×1016 years and 3.2×1014 years, respectively.
Place of Birth
Platinum nugget, Conder deposit
Platinum is one of the rarest metals: its average content in the earth's crust (clarke) is 5⋅10−7% by mass. Even the so-called native platinum is an alloy containing from 75 to 92 percent platinum, up to 20 percent iron, as well as iridium, palladium, rhodium, osmium, and less often copper and nickel.
The bulk of platinum deposits (more than 90%) are located in the subsoil of five countries. These countries include South Africa (Bushveld complex), USA, Russia, Zimbabwe, China.
In Russia, the main deposits of platinum group metals are: Oktyabrskoye, Talnakhskoye and Norilsk-1 sulfide-copper-nickel in the Krasnoyarsk Territory in the Norilsk region (more than 99% of explored and more than 94% of estimated Russian reserves), Fedorova Tundra (Bolshoi Ikhtegipakhk site) sulfide- copper-nickel in the Murmansk region, as well as alluvial deposits Konder in the Khabarovsk Territory, Levtyrinyvayam in the Kamchatka Territory, the Lobva and Vyisko-Isovskoye rivers in the Sverdlovsk Region. The largest platinum nugget found in Russia is the “Ural Giant” weighing 7860.5 g, discovered in 1904 at the Isovsky mine; currently stored in the Diamond Fund.
Where and how is it mined?
According to the theory, the process of formation of platinum is the same as that of gold - from interstellar dust as a result of a cosmic explosion.
She found herself in the core of the planet, then rose to the surface by earthquakes:
- It is found in igneous igneous rocks.
- Platinum veins are found in river beds, next to other minerals.
- Platinum in nature is usually found together with platinum group metals, iron, copper, and nickel. Most often these are large nuggets; cube crystals are rare.
Today the world leader in the extraction and supply of raw materials is South Africa. Russia is in the top five.
The methods for extracting platinum raw materials are similar to gold - washing and extraction from other ores. Then treatment using the refining method (removal of impurities) - double exposure to “regia vodka”, calcination.
Dissolving platinum in hot aqua regia
Receipt
Native platinum is mined at mines (see the article Noble Metals for more details); placer deposits of platinum are less rich, which are explored mainly by the method of spot testing.
The production of platinum in powder form began in 1805 by the English scientist W. H. Wollaston from South American ore.
Today, platinum is obtained from a concentrate of platinum metals. The concentrate is dissolved in aqua regia, after which ethanol and sugar syrup are added to remove excess HNO3. In this case, iridium and palladium are reduced to Ir3+ and Pd2+. The subsequent addition of ammonium chloride produces ammonium hexachloroplatinate (IV) (NH4)2PtCl6. The dried sediment is calcined at 800–1000 °C:
3(NH4)2[PtCl6] →T 2N2↑ + 2NH3↑ + 18HCl + 3Pt
The sponge platinum thus obtained is subjected to further purification by repeated dissolution in aqua regia, precipitation of (NH4)2PtCl6 and calcination of the residue. The purified sponge platinum is then melted into ingots. When reducing solutions of platinum salts by chemical or electrochemical methods, finely dispersed platinum is obtained - platinum black.
Application of platinum
Platinum raw materials are divided into three areas of application (%):
- industry, technology – 60;
- jewelry – 30;
- financial sector – 10.
Platinum fragments are on the highest awards of the USSR: the orders of Lenin, “Victory”, Suvorov and Ushakov (both 1st degree).
Platinum image of Lenin on the Order of Lenin
The properties of platinum have made it indispensable in practical applications.
Industrial complex
Noble metal helps make human life easier:
- From a third to a half of the metal volume is taken by the auto industry. Thanks to platinum catalysts, almost all emissions (exhaust gases) are neutralized.
- Oil distillation, production of silicone products, electrical and aircraft parts, and special types of glass.
- Dental and medical instruments, means for the treatment of cardiac diseases (platinum-iridium electrodes), oncological diseases.
- Artificial fertilizers for the agricultural complex.
Standards of measurement are made from platinum. In France it is a meter and a kilogram, in Russia it is a kilogram (a platinum-iridium cylinder with a height and a diameter of 39 mm; stored in St. Petersburg).
Jewelry
Platinum is expensive, but is in demand by jewelers and their clients due to its obvious advantages:
- All products are elegant, with a noble, discreet shine.
- Diamonds in such a setting look even more expensive and hold more securely.
- White color goes well with precious stones of all shades.
- Platinum jewelry wears out little and is therefore suitable for constant wear.
- Unlike gold, it is hypoallergenic. That is, it suits everyone.
The only “disadvantage” of platinum is the price. It is several times higher than gold . It’s all the more prestigious to buy at least a ring or stud earrings made of royal metal.
Financial sector
Platinum is used to make bank bars, collectible coins, and medals.
Platinum coin of 1835 with a face value of 12 rubles
You can also buy or sell “virtual” platinum on the exchange.
Platinum samples
Platinum is durable, but its alloys are used in jewelry. Therefore, metal is subject to a standard system - metric and carat.
Unlike other metals, platinum is not coated with anything, so the classification is carried out by type of sample.
The sample number shows how much pure metal the alloy or product contains.
For example, 1 kg of 850-grade alloy is 850 g of pure platinum and 150 g of impurities.
Slovakia and the Czech Republic have their own sampling system (indicated in the table as SC).
The 800th sample is present here (marked as 4).
Characteristics of the main platinum samples:
| Sample (metric/carat/SP) | Impurities (ligatures) | Scope of application |
| 850/20/3 | Iridium, tungsten, cobalt or copper. | There is little demand among jewelers; it is often used for cutlery and small plastic items. |
| 900/22/2 | Ruthenium, iridium | Jewelry |
| 950/23/1 | Palladium, ruthenium, iridium | Decorations, small plastic items, deluxe cutlery. |
| 999/24/0 | None | Bank bullion, in industry – special purpose tools |
Sample 999 has special features. The content of precious metal in the alloy is measured to hundredths of a percent - from 99.95 to 99.99%.
The platinum standard is “five nines”, 99.999% platinum.
How to determine authenticity
They offer silver or white gold under the guise of platinum.
You can determine the authenticity of a product at home:
- A product made of platinum is heavier than gold, especially silver of comparable parameters.
- Place the silver item and the sample next to it. Even high-grade silver gives off a grayish tint and “coldness.” The platinum composition has a pure warm white luster.
There are other differences between platinum and white gold:
- Platinum rings, earrings, even necklaces and bracelets are not large or massive - the metal is too expensive.
- At normal temperatures, it is not affected by hydrogen peroxide, iodine solution, or acetic acid. They will not leave marks on the surface.
- Under the flame of a candle, lighter, or household gas stove, it will heat up, but will not change in appearance. Gold may soften.
- You can hold the sample in your hands: the royal metal takes longer to heat up than gold, especially silver.
- A pawn shop worker or jeweler is guaranteed to be able to distinguish the original.
Platinum looks almost like white gold, so many are sure that they are the same thing. However, white gold is an alloy of metals, and platinum is an independent element.
Origin
Found in primary deposits or placers. The former are of two types: a) inclusions or local impregnations of native platinum in ultrabasic igneous rocks (dunites) associated with chromite and b) copper-nickel sulfide accumulations in basic (norite) rocks. In basic magmas, at temperatures of 1300–1500° C, sulfides separate from the silicate melt. Chromite, osmium, iridium and platinum separate at higher temperatures; palladium is at the same or lower temperature and is thus captured in the sulfide melt. Therefore, native platinum and osmiride are the main minerals in the first type of deposits. The second type contains a lot of palladium and platinum, often found in the form of arsenide (sperrylite).
Contact-metamorphic platinum deposits, as well as quartz veins containing platinum, are also known.
Platinum group minerals are mostly found in typical igneous deposits genetically related to ultramafic igneous rocks. These minerals are among the last to be released in ore bodies (after silicates and oxides) at moments corresponding to the hydrothermal stage of the magmatic process.
Platinum minerals poor in palladium (polyxene, platinum iridium, etc.) are found in deposits among dunites - olivine feldspathic rocks rich in magnesia and poor in silica. Moreover, paragenetically they are extremely closely related to chrome spinels - oxides of complex composition: (Fe,Mg)(Cr,Al,Fe)2O4.
Palladium and nickel-palladide platinum are predominantly common in basic igneous rocks (norites, gabbro-norites) and are usually associated with sulfides: pyrrhotite (Fe1-XS), chalcopyrite (CuFeS2) and pentlandite - (Fe,Ni)9S8.
Under exogenous conditions, in the process of destruction of bedrock deposits and rocks, platinum placers are formed. Most of the minerals of the subgroup are chemically resistant under these conditions.
How to store and care
In order for platinum products to maintain their original premium condition, simple care is sufficient:
- Jewelry (especially rings and bracelets) is removed before household work (washing, cleaning, cooking).
- They are protected from moisture and household chemicals, especially aggressive ones.
- It should be stored in a separate, tightly closed box.
Tarnish or dirt can be removed by immersing the item in a warm soapy solution for a couple of hours. Then rinse with water and dry with a soft cloth.
If possible, purchase a cleaning product for platinum products.
If the jewelry contains diamonds or other stones, it should be sent for professional cleaning.
Why is platinum so expensive?
Platinum is a premium metal, a marker of luxury.
This is natural:
- It is found on Earth 30 times less often than gold.
- To obtain 100 g of pure platinum raw materials, it is necessary to process 30 tons of ore. Gold is four times less.
- Only Russia, South Africa, the USA, China, and Zimbabwe have industrial-scale platinum deposits.
- World reserves are estimated in tons (not thousands of tons like gold).
World exchange prices for platinum exceed gold and also fluctuate depending on global market conditions. For example, at the end of December 2021, 1 gram costs 2.4 thousand rubles.
Extraction methods
The production process consists of three stages:
- Ore mining.
- Enrichment.
- Obtaining precious metal of high purity.
Extracting platinum from the earth's interior is a labor-intensive and expensive task. To extract 1 ounce (31.1 grams) of precious metal, more than 10 tons of ore are processed.
There are two ways to get it:
- open;
- underground.
The first option is suitable for placer deposits formed as a result of the destruction of primary rock. It involves the use of quarry equipment, dredges, and hydromechanical means.
Primary deposits and buried placers are mined underground. Shafts are dug, holes are manually drilled into them, and explosives are placed. Miners lift pieces of rock that break off to the surface for further processing. Today, this version of metal mining is significantly mechanized, but it cannot be done without manual labor.