Not everyone knows, but diamond and graphite are two forms of the same substance. These minerals are completely different from each other in hardness and in the characteristics of refraction and reflection of light. Moreover, the differences are quite significant. Diamond is the hardest mineral in the world, on the Mohs scale it represents a standard of 10, while the hardness of graphite on this scale is only 2. Thus, diamond and graphite are simultaneously the most similar and dissimilar substances in the world.
Crystal lattices of diamond and graphite
Each of them comes from carbon, which, in turn, is the most abundant element in the biosphere. It is present both in the atmosphere and in water, in biological objects. In the ground it is present in the composition of oil, gas, peat, and so on. It is also found as deposits of graphite and diamond.
Most carbon is found in organisms. Moreover, none of them can do without it. And the origin of this mineral in other parts of the planet is precisely explained by the presence of living organisms once there.
Much controversy surrounds the question of where graphite and diamonds came from, because it is not enough for there to be carbon alone; it is also necessary that certain conditions be met under which this chemical element takes on a new structure. It is believed that the origin of graphite is metamorphic, and diamonds are igneous. This means that the formation of diamonds on the planet is accompanied by complex physical processes, most likely in the deep layers of the earth during combustion and explosions in the presence of oxygen. Scientists suggest that methane is also involved in this process, but no one knows for sure.
The concept of allotropy
The concept of “Allotropy” has ancient Greek roots: αλλος - other, τροπος - property. Allotropy is the existence of two or more simple substances of the same chemical element. The concept of allotropy was introduced into science by J. Berzelius in 1841 to designate different forms of existence of elements.
The phenomenon of allotropy implies the possibility of creating a certain amount of different substances from the same element. For example, oxygen and ozone contain only oxygen in their composition. The question of how this is even possible has interested many people for a long period of time. Today, scientists can easily explain all the features of this process.
Not all elements are capable of forming several different simple substances. This ability directly depends on the structure of the molecules. Most often, this phenomenon is observed in elements that have variable oxidation states.
This applies to groups such as:
- non-metals;
- semimetals;
- noble gases;
- halogens.
The causes of allotropy can be of several types. The most likely of them, scientists include the following factors:
- Different numbers of atoms required to form one molecule.
- A different order of conjugation of atoms into one molecule.
- Parallels between electron spins.
- A type of crystal lattice.
In order to clearly understand how the phenomenon of allotropy can exist, it is necessary to consider several of the most remarkable examples that are widely found in nature.
Examples of allotropy:
When metals are heated during the transformation process, heat is absorbed, and the change in lattice structure occurs at the same temperature. Many metals are subject to allotropic modifications, for example, titanium, iron, tin, etc. When heated to +1390 ºC, iron is characterized by a face-centered lattice. An increase in temperature to +1540 ºС leads to a restructuring to a centered cubic structure.
Optical characteristics
The uniqueness and such a high price of diamonds on the jewelry market is due to two factors
: hardness and optical characteristics. No mineral on earth has such indicators. The refractive index is 2.41. This indicator is based on the deflection of a light beam in another environment where there is a sharp change in the speed of light.
Coupled with the high dispersion value, it has such a bright glow, and if you shine a beam of light on it, it will be reflected in all the colors of the rainbow. No mineral, even artificially created, has such capabilities.
Allotropic modifications of carbon
Allotropic modifications of carbon differ most radically from each other in their properties, from soft to hard, etc.
Discovery history:
Carbon in the form of coal, soot and soot has been known to man since time immemorial; about 100 thousand years ago, when our ancestors mastered fire. Probably, very early people became acquainted with allotropic changes in carbon - diamond and graphite, as well as fossil coal. It is not surprising that the combustion of carbon-containing substances was one of the first chemical processes to interest man.
The element was fire, the phenomenon accompanying combustion; In ancient teachings about the elements, fire usually appears as one of the elements. At the turn of the XVII-XVIII centuries. The phlogiston theory arose, put forward by Becher and Stahl. This theory recognized the presence in each combustible body of a special elementary substance - a weightless fluid-phlogiston, which evaporates during the combustion process.
Since when a large amount of coal is burned, only a little ash remains, phlogistics believed that coal was almost pure phlogiston. This is what explained, in particular, the “phlogisticating” effect of coal—its ability to restore metals from “limes” and ores. Later phlogistics, Reaumur, Bergman and others, already began to understand that coal is an elementary substance. However, “clean coal” was first recognized as such by Lavoisier, who studied the process of combustion of coal and other substances in air and oxygen.
In the book “Method of Chemical Nomenclature” by Guiton de Morveau, Lavoisier and Fourcroix (1787), the name “carbon” appeared instead of the French “pure coal”. Under the same name, carbon appears in the “Table of Simple Bodies” in Lavoisier’s “Elementary Textbook of Chemistry.” In 1791, the English chemist Tennant was the first to obtain free carbon; he passed phosphorus vapor over calcined chalk, resulting in the formation of calcium phosphate and carbon. It has been known for a long time that diamond burns without leaving a residue when heated strongly.
Back in 1751, the French king Francis I agreed to provide diamond and ruby for burning experiments. It turned out that only diamond burns, and ruby (aluminum oxide with an admixture of chromium) can withstand prolonged heating at the focus of the ignition lens without damage. Lavoisier carried out a new experiment on burning diamonds using a large incendiary machine and came to the conclusion that diamond is crystalline carbon.
The second allotrope of carbon - graphite in the alchemical period was considered a modified lead luster and was called plumbago; It was only in 1740 that Pott discovered the absence of any lead impurity in graphite. Scheele studied graphite (1779) and, being a phlogistician, considered it a special kind of sulfur body, a special mineral coal containing bound “aerial acid” (CO2) and a large amount of phlogiston. Twenty years later, Guiton de Morveau turned diamond into graphite and then into carbonic acid by careful heating.
The international name Carboneum comes from the Latin carbo (coal). This word is of very ancient origin. It is compared with cremare - to burn; root car, cal, Russian gar, gal and gol, Sanskrit cra means to boil, cook. The word “carbo” is also associated with the names of carbon in other European languages (carbon, charbone, etc.). The German Kohlenstoff comes from Kohle - coal.
Old Russian ugorati, or ugarati (to burn, scorch) has the root gar, or mountains, with a possible transition to gol; coal in Old Russian yugal, or coal, of the same origin. The word diamond comes from the ancient Greek - indestructible, unyielding, hard, and graphite from the Greek - I write. At the beginning of the 19th century. The old word coal in Russian chemical literature was sometimes replaced by the word “carbonate” (Shere, 1807; Severgin, 1815); Since 1824, Soloviev introduced the name carbon.
Definition
Diamond
- a mineral based on carbon. It is characterized by metastability, that is, the ability to exist in an unchanged form under normal conditions for an indefinitely long time. Placing diamond in specific conditions, for example in a vacuum at an elevated temperature, leads to its transition to graphite.
Diamond
Graphite
– a mineral that acts as a modification of carbon. During friction, scales are separated from the total mass of the substance. The most famous use of graphite is making pencil leads from it.
Graphite
Diamond
Carbon has many allotropic modifications. The main ones are graphite, diamond, carbine .
They differ from each other in physical, chemical properties, and crystal lattice structure. The hardest modification, diamond, is used for the manufacture of industrial tools. According to modern theoretical concepts, nothing in the world can be harder than diamond - such is its crystal lattice. We can say that diamond is the hardest mineral in the world. Diamond melts at high temperatures, from 3700 to 4 thousand degrees. But even earlier, at 850 degrees, it begins to burn, and without air access, when it reaches half the melting temperature, it turns into graphite.
Mining history
Diamonds were not always precious stones that had value and a scale of value. In nature, this pebble is unattractive - a simple rough piece of glass. It is the cut that gives it its value. Everything changed in the 19th century, when diamond seekers settled on the farm of the de Beers brothers in South Africa near the modern city of Kimberley. There were a lot of stones on these lands. Their real industrial production became associated with the name of Cecil Rhodes.
Cecil Rhodes became a monopolist in the diamond market, which was facilitated by the Rothschilds, and diamonds became a consumer product available not only to kings. De Beers' monopoly position was shaken only in the middle of the 20th century thanks to antitrust laws in the USA and the beginning of mass production in countries where there were no opportunities to capture the market in principle - for example, in the USSR. There are several ways to cut diamonds, in which they best demonstrate their properties of color play. The original shape of the stone also plays a role, since the cutter tries to reduce its losses to a minimum.
The most common diamond shapes are:
- round, 57 edges;
- oval;
- "pear";
- "marquise";
- radiant;
- square;
- "princess".
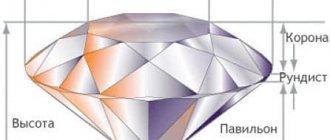
Diamond Structure:
One carbon atom is surrounded by four more atoms in the form of a tetrahedral triangle or pyramid. Each atom is the same distance from each other. The bonds between atoms are very strong, which is why diamond is so hard and durable. Another property of diamond is that it can conduct light, unlike graphite.
Mineral deposits
Diamonds originate at a depth of 100 km and at a temperature of 1300 degrees. Kimberlite magma, which forms kimberlite pipes, comes into action through explosions. It is these pipes that represent the primary diamond deposits. The first such pipe was discovered in the African province of Kimberley, where its name came from.
The most famous deposits are located in India, Russia and South Africa. Primary deposits account for 80% of all mined diamonds.
To find a diamond in nature, X-rays are used. Most of the stones that are found are unsuitable for jewelry production, as they have a significant number of defects, including cracks, inclusions, foreign fluorescent shades, and so on. Therefore, their use is technical. Such stones are divided into three categories:
- board - stones with a zonal structure;
- ballas - stones that are round or pear-shaped;
- carbonado - black diamond.
Diamonds that are large in size and have outstanding characteristics tend to get their name. In addition, the high cost of the stone makes it desirable for many, which guarantees a “bloody history.”
Graphite is formed by alteration of sedimentary rocks. In Mexico and Madagascar you can find low quality graphite ore. The most famous deposits are in Krasnodar and Ukraine.
Graphite
Graphite is a mineral from the class of native elements, one of the allotropic modifications of carbon. The structure is layered. The layers of the crystal lattice can be differently positioned relative to each other, forming a number of polytypes, with symmetry from hexagonal system (dihexagonal-dipyramidal) to trigonal (ditrigonal-scalenohedral).
The layers are slightly wavy, almost flat, and consist of hexagonal layers of carbon atoms.
Mining history:
In the 60s of the 16th century in England. Local shepherds, who found deposits of a strange black-shiny material, at first mistook it for lead, but, realizing that they couldn’t cast bullets from it, they began to break off pieces of black stone and mark their sheep with it. The new material soon attracted the attention of artists and merchants, who quickly established trade in thin plates and pieces of graphite on the English streets. Of course, it was very inconvenient to use - your hands got dirty! I had to wrap the graphite with rope, paper, or even simply press it between boards. This is how the first pencils in a wooden case appeared.
Graphite structure:
Physical properties in graphite vary greatly in directions - perpendicular and parallel to the layers of carbon atoms.
When heated without air access, graphite does not undergo any changes up to 3700°C. At the specified temperature, it sublimes without melting.
Artificial graphite is produced from the best grades of coal at 3000°C in electric furnaces without air access.
Graphite is thermodynamically stable over a wide range of temperatures and pressures, so it is accepted as the standard state of carbon. The density of graphite is 2.265 g/cm3.
There are two known forms of graphite: alpha graphite (has a hexagonal structure and crystal lattice) and beta graphite (has a rhombohedral structure and crystal lattice). In α-graphite, half of the atoms of each layer are located above and below the centers of the hexagon, and in β-graphite, every fourth layer repeats the first.
Alpha graphite can be converted to beta form by mechanical processing. The beta form transforms into the alpha form when graphite is heated above 1300 °C.
Medicine
In medicine, industrial diamonds are a necessary element in the manufacturing process of surgical scalpels, scissors, clamps, and dental equipment. A special coating applied to the blade guarantees a more precise and neat cut. Such instruments are indispensable during complex operations on the spinal cord and brain.
Another area of application is medical lasers. The crystal is built into a complex device as a conductor. Laser operations are increasingly used in modern surgery, as they are less traumatic and require a shorter rehabilitation period for patients.
Until the mid-50s of the last century, mainly technical types of diamonds were used in industry. But after 1953 they were replaced by artificial analogues with exactly the same technical characteristics.
Fullerene
Fullerene , buckyball, or buckyball is a molecular compound that belongs to the class of allotropic forms of carbon and is a convex closed polyhedron composed of an even number of three coordinated carbon atoms.
The unique structure of fullerenes determines their unique physical and chemical properties. In combination with other substances, they make it possible to obtain materials with fundamentally new properties. Mining history:
The discovery of the fullerene resulted from experiments by Smalley and Croteau with an instrument that Smalley had invented to study molecules and atomic clusters. Kroteau was interested in Smalley's proposed laser evaporation technique. With its help, he intended to test his theory about the behavior of carbon in interstellar space. Croteau believed that carbon-rich red giants were capable of emitting complex carbon compounds that could be detected by radio telescopes.
Fullerene structure:
Atom bond Fullerene is a new allotropic form of carbon. Fullerene molecules consist of 60.70 atoms forming a sphere. Crystalline fullerenes are semiconductors. The variety of physicochemical and structural properties of compounds based on fullerenes allows us to talk about the chemistry of fullerenes as a new promising direction in organic chemistry.
Carbon atoms are located at the vertices of regular hexagons and pentagons that make up the surface of a sphere or ellipsoid. The most symmetrical and most fully studied member of the fullerene family is fullerene (C60), in which the carbon atoms form a truncated icosahedron consisting of 20 hexagons and 12 pentagons and resembling a soccer ball.
The next most common is the C70 fullerene, which differs from the C60 fullerene by the insertion of a belt of 10 carbon atoms into the equatorial region of C60, as a result of which the C70 molecule is elongated and resembles a rugby ball in shape. The so-called higher fullerenes, containing a larger number of carbon atoms (up to 400), are formed in much smaller quantities and often have a rather complex isomeric composition. Among the most studied higher fullerenes are Cn, n=74, 76, 78, 80, 82 and 84.
Jewelry craftsmanship
The most famous industry where diamonds are involved is the jewelry industry. Moreover, here the mineral is used both as an object and as a means of processing it. Diamond-coated equipment is a necessary condition for high-quality cutting. It is with its help that a precious diamond is born from a modified form of carbon - one of the most expensive gems in the world.
This stone is often the centerpiece of imperial and royal treasures. Both the crown of the Russian emperors and the crown of the British Empire are decorated with it, and it is inserted into sceptres, royal staffs and orbs. Owning jewelry with diamonds emphasizes the status and position in society and demonstrates the financial condition of the owner. Jewelry with this stone often became family heirlooms and was passed down from generation to generation.
The value of diamond jewelry does not decrease over time, but only increases in price.
Physical properties
Physical properties. Carbon exists in a variety of allotropes with very diverse physical properties. The variety of modifications is due to the ability of carbon to form chemical bonds of different types.
Therefore, different modifications exhibit very different physical properties: a very hard substance, a soft substance that conducts electric current, and many others.
Diamond:
Phys. properties. Diamond and graphite differ sharply in physical properties. Properties. Diamonds are transparent crystals, very hard. Hardness is explained by the structure of its crystal lattice. Graphite is a soft dark gray substance with a Me shine.
Graphite:
Physical properties of graphite. – a soft black substance made of easily exfoliated crystals, – conducts electric current, – high-purity graphite is used in nuclear reactors as a neutron moderator. - the melting point at elevated pressure is 3527° C. - At normal pressure, graphite sublimates at 3780° C.
Fullerene:
Physical properties of fullerene Condensed systems consisting of fullerene molecules are called fullerites. The most studied system of this kind is the C60 crystal, less so is the system of crystalline C70. Studies of crystals of higher fullerenes are hampered by the complexity of their preparation.
Therapeutic effect
Homeopaths were the first to appreciate graphite. They found that the mineral is suitable for the treatment of skin pathologies (eczema, psoriasis, lichen, etc.).
Today the list has been expanded:
- Metabolic disease.
- Malfunction of the thyroid gland.
- Respiratory tract diseases (rhinitis, bronchial asthma).
- Gastrointestinal problems (gastritis, gastric ulcer, duodenal ulcer, colitis).
- Women's ailments (amenorrhea, chronic inflammation of the ovaries, mastopathy).
- Conjunctivitis, cataracts, stye.
The mineral also “supervises” emotional health. It is prescribed for morning headaches, neurasthenia, apathy, and depression.
Sources
- https://vseprokamni.ru/vidy/organicheskie/ximicheskaya-formula-grafita.html
- https://FB.ru/article/351885/chto-takoe-grifel-iv-ch-m-zaklyuchayutsya-ego-unikalnyie-svoystva-dlya-risovaniya
- https://mineralpro.ru/minerals/graphite/
- https://www.syl.ru/article/307195/formula-grafita-allotropiya-ugleroda
- https://FB.ru/article/277065/grafit-plotnost-svoystva-osobennosti-primeneniya-i-vidyi
- https://vseprokamni.ru/svoistva/kristallicheskaja-reshetka-grafita.html
- https://crystal-wow.ru/kamni/fizicheskie-svojstva-grafita-tablica.html
- https://vseprokamni.ru/svoistva/fizicheskie-i-himicheskie-svojstva-granita.html
- https://natrukodel.ru/prochie/himicheskaya-formula-grafita
- https://abc-24.info/prostoj-karandash-istoriya/
- https://FB.ru/article/236131/allotropnyie-veschestva-almaz-i-grafit-formula-grafita-i-almaza
Chemical properties
Diamond:
At ordinary temperatures, carbon is inactive. It can be both a reducing agent and an oxidizing agent. As a reducing agent: Burns in air.
Graphite:
Chemical properties. It forms inclusion compounds with many substances (alkali metals, salts). Reacts at high temperatures with air, burning to carbon dioxide.
Fullerene:
Reduction, nucleophilic addition, cycloaddition, regiochemical multiple addition, halogenation, modification of fullerenes, cluster hydrogenation, addition of radicals, formation of complexes, transition metal oxidation and reactions with electrophilic reagents.
Practical part
Application of allotropic modifications of carbon. Diamond - in industry: it is used to make knives, drills, cutters; in jewelry making. The future is the development of microelectronics on diamond substrates. Graphite – for the manufacture of melting crucibles, electrodes; plastic filler; neutron moderator in nuclear reactors; component of the composition for the manufacture of leads for black graphite pencils (mixed with kaolin) Fullerene - in batteries and electric batteries (fullerene additives); pharmacology (HIV treatment); Solar cells; fire retardant paints.
New allotropic modification:
On August 15, 2021, a team of scientists from IBM and the University of Oxford published a paper presenting data on the successful synthesis of the cyclo[18]carbon molecule. Previously, the existence of cyclocarbons was considered only hypothetical, but now C18 is a representative of a new allotropic modification of carbon.
C18 was obtained by removing carbon monoxide from the C24O6 molecule by the principle of atomic force microscopy on a two-layer sodium chloride surface at a temperature of 5°K (-268.15°C). According to scientists, cyclocarbons may prove useful in computer technology as an extremely energy-efficient computer logic device. In addition, the creation of C18 opens the way for the synthesis of other carbon allotropes, but for now, an in-depth study of the properties of the new molecule remains.
Conclusion
Carbon is the substance with the largest number of allotropic modifications. The project considers allotropic modifications of diamond, graphite, and fullerene. Carbon and its allotropic modifications are of great practical importance in human life and industry.
Most of the things in our lives we received thanks to allotropic modifications, for example, a drill, black graphite pencils and batteries.
Thanks to this project, we learned how and who helped us improve our lives.
Based on all the work done, we can draw the following conclusions:
- Firstly, I studied and examined in detail the types of allotropy.
- Secondly, I found out who discovered allotropy and where it is used.
- Thirdly, I made a stand that clearly shows what allotropy looks like.
Areas of application
The properties of graphite allow it to be used in everyday life and in industry. Due to its high fire resistance and electrical conductivity, it is used in metallurgy for the production of molds and ladles . Foundries use the powder as a lubricant.
In addition, graphite is added to refractory bricks, grinding and polishing solutions. The most famous use is in the manufacture of drawing pencils. Even the nuclear energy industry uses it at its facilities.
Graphite paint is considered one of the highest quality paints. With the help of such a mixture, it is possible to provide effective protection of cast iron, aluminum, wood and concrete products from corrosion.
In the medical field, graphite is used in the treatment of skin diseases, which are the result of a variety of internal disorders. This element also prevents the formation of scars and adhesions after serious inflammation and has a positive effect on metabolic processes. That is why it is added to many medicines.
How are graphite rods produced?
The most important operation in the production of rods is grinding graphite. The smaller the particles, the smoother the rods will be, the better they will write. Mechanical grinding methods require a long time, and still do not produce sufficiently fine particles. Therefore, mechanochemical methods are used; a surfactant is introduced into a vibration or jet mill, which wets the crushed particles and prevents them from sticking together again. As a result, the sizes of graphite particles are negligible - about one micron.
For clarity, I would like to compare them with something known, but even the finest powder is coarser than pencil graphite. But there is no need to grind the clay, it is already fine enough: its particles have submicroscopic sizes. Graphite is mixed with clay. The hardness of the future rod depends on the ratio in which it is mixed. The hardest will come from pure clay, the softest from pure graphite. Of course, both of these extremes are absurd. It’s just that there is very little graphite in 6T, while there is a lot of it in 6M.
A mixture of graphite and clay is forced through a nozzle and a continuous black snake is automatically cut into individual rods. And then they send it to the oven. Pencil clays are made from kaolinite. At high temperatures, water is released from it and a dense polymer is formed. Therefore, the rod becomes strong, water-resistant and more elastic, its hardness increases by one to one and a half grades.
But due to the evaporation of water, the rod after firing is literally saturated with tiny interconnected pores; the line drawn by it is intermittent and uneven. You have to impregnate the fired rod with waxes - suitable, Japanese, carnauba - or wax-like substances like stearin. By the way, such substances also improve the adhesion of what is written to paper and reduce friction when writing.